- Shanghai Zhongshen International Trade Co., Ltd. - Two decades of trade agency expertise.
- Service Hotline: 139 1787 2118
Medical EquipmentThe medical device industry, characterized by high - tech content, high investment, high risks, but also high returns, has received widespread attention globally. In recent years, with the implementation of Chinas centralized procurement policy, domestic medical device enterprises are facing fierce price competition and increasing quality requirements. This not only intensifies homogeneous competition but also squeezes the profit margins of enterprises. Against this backdrop, Chinese medical device enterprises have begun to turn their attention to foreign markets.
The global demand for medical devices is huge. Especially with the global population aging and the improvement of medical standards, this demand is still growing continuously. There is a wide variety of medical devices, covering aspects from diagnostic and monitoring equipment to clinical laboratory equipment, and then to rehabilitation and nursing equipment. To gain a foothold in this vast market, domestic enterprises need to carry out technological innovation to adapt to different medical needs.
In foreign markets, the import classification of medical device products is usually carried out according to their use, technical characteristics, and risk levels. According to the risk levels, these products are divided into Class I, Class II, and Class III. Different types of products need to go through different certification and registration procedures. For Chinese medical device enterprises that are new to overseas markets, they can start with relatively low - threshold segments such as medical consumables, low - risk medical devices, and non - regulated medical devices.
In foreign markets, products such as high - tech medical devices, diagnostic equipment, rehabilitation and assistive devices, and medical consumables are more popular. Especially high - tech medical devices and diagnostic equipment usually receive more attention and have greater demand. During the process of going global, Chinese enterprises currently tend to adopt the registration model, that is, registering products in overseas markets to comply with local regulations and standards, obtaining sales licenses, and entering the local market for sales. This approach can enhance the products market competitiveness, market access threshold, and the enterprises brand reputation, etc.
According to statistical analysis, from 2016 to October 2023, the number of registrations of Chinese manufacturers overseas showed an increasing trend year by year. The number of registrations in 2022 was nearly double that in 2021. This indicates that Chinese enterprises are accelerating their pace of going global. In terms of overseas region selection, Chinese enterprises usually give priority to regions with large medical device markets and high medical standards, such as Europe, the United States, Japan, Australia, and Canada.
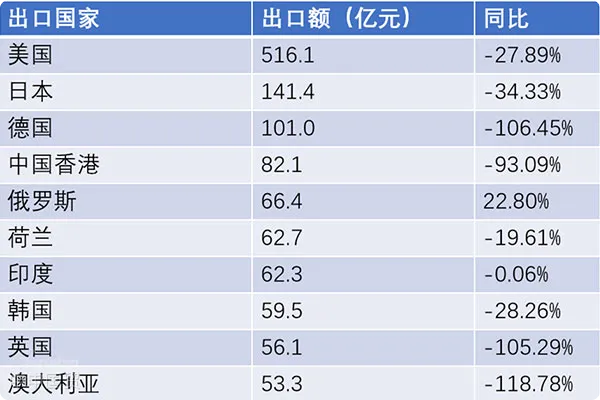
However, the export of the medical device industry has also experienced ups and downs. In recent years, under the influence of the global pandemic, the export of medical devices from China has shown a sharp increase. But as the pandemic becomes normalized, the global order volume for epidemic prevention products has been declining, and the trade of medical devices has experienced huge fluctuations. From 2016 to 2019, the export of Chinese medical devices showed a steady upward trend. During the pandemic in 2020, the export growth rate was as high as 13.78%. But in 2022, as the market returned to normal, the export growth rate dropped to - 2.34%.import and exportThe United States is the largest export market for Chinese medical devices, accounting for more than 35% of the market share. In addition to the United States, Germany, Japan, the United Kingdom, the Netherlands, Australia, South Korea, and Hong Kong, China are also major export markets. From January to October 2023, the United States was still the country with the largest export value of Chinese medical devices, reaching 51.61 billion yuan.
Despite many opportunities, Chinese medical device enterprises also face many challenges during the process of going global. These include unfamiliarity with local regulations and certification processes, difficulty in finding suitable local agents and distributors, lack of experience in dealing with certification agencies, and difficulty in obtaining accurate local market data.
The Current Situation and Trends of Chinese Medical Devices Exported Overseas | Shanghai Import/Export Agent
Related Recommendations
Category case
Contact Us
Email: service@sh-zhongshen.com
Related Recommendations
Contact via WeChat

? 2025. All Rights Reserved. Shanghai ICP No. 2023007705-2  PSB Record: Shanghai No.31011502009912
PSB Record: Shanghai No.31011502009912